|
7/23/2023 0 Comments Pcl3 electron domain geometry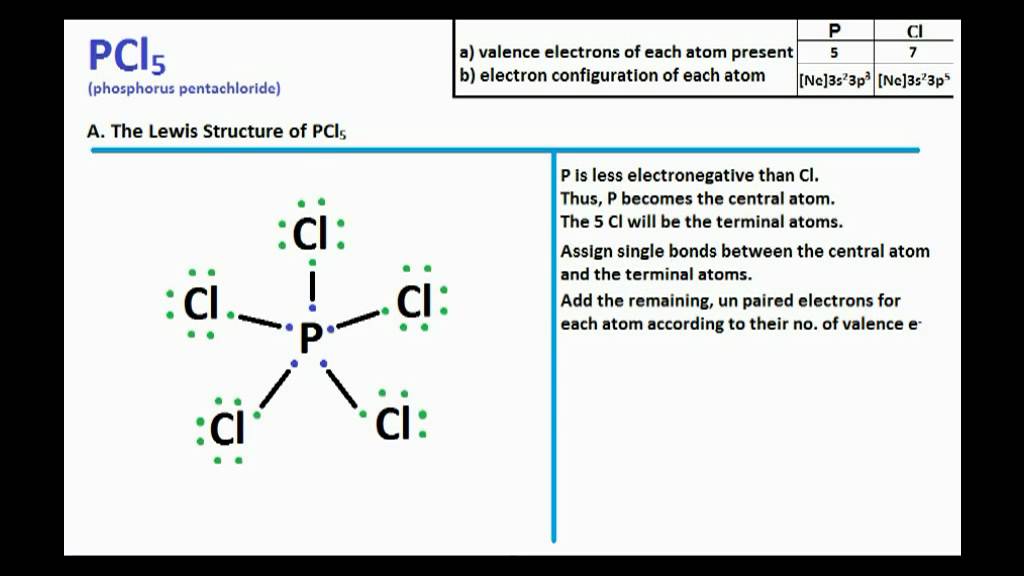
WebHow many bonding pairs and how many lone pairs are there in PCl 3? Group of answer choices. How many lone pairs are in PCl3? – how many lone pairs are in pcl3 mean NOCl Lewis structure, Molecular Geometry, Shape and Hybridization What is the number of electron domains? - De Kooktips Number of lone pairs around phosphorus in PCl5, PCl4^ + and … How many lone pairs are in pcl3 mean Answered: How many of the following three choices… bartleby The Phosphorus atom (P) is at the center and it is … Lewis structure of PCl3 contains three single bonds between the Phosphorus (P) atom and each Chlorine (Cl) atom.PCl 4+: + sign signifies loss of 1 electron, rest 4 involved in bonding. How many lone pairs are in pcl3 mean WebNumber of lone pairs around phosphorus in PCl 5,PCl 4+ and PCl 6− are respectively A 0,1,2 B 0,0,0 C 1,2,3 D 0,2,1 Medium Solution Verified by Toppr Correct option is B) P atom has 5 valence electrons, PCl 5, all 5 electrons involed in bonding. 
We often need to know the the number of lone pairs of electrons in a molecule like I3- (as well as electrons … WebThere are three lone pairs of electrons on the central I atom in I3. PCl3 Molecular Geometry - Science Education and Tutorials Solved In the Lewis structure of PCl3, how many bonds … PCl3 Lewis structure - Learnool how many lone pairs are in pcl3 mean Number of Lone Pairs and Bonding Pairs for I3 - YouTube For the Lewis structure above, how many lone pairs are on the P in PCI3 A.) Two B.) four C.) one D.) - Chemistry College answered For the ….Solved How many bonding pairs and how many lone pairs are - Chegg WebQuestion 5 (2 points) When drawing the Lewis Dot Structure of PCl3, how many lone pairs are there in the whole molecule? 3 lone pairs 1 lone pair 10 lone pairs O lone pairs This problem has been solved! You'll get a detailed solution from a subject matter expert that helps you learn core concepts. The IUPAC definition of oxidation state requires the oxygen have a -2 oxidation state except in peroxides. There is resonance between various Lewis structures as: : C ⊖ ≡ O ⊕: : C = O :: : C ⊕ − O ⊖ ::: The term "valence" is not used much nowadays, and "oxidation state" is used but has a different meaning. Sulfor dioxide: Lewis dot structure for SO2 (video) Khan Academy In PC 1 3 how many atoms bond to the central atom and how many lone pairs are there in it? In PCl3 there are three bonds to the central atom (P) and one lone … photography 48858.Solved Question 5 (2 points) When drawing the Lewis Dot - Chegg WebAll bond angles are 90° linear AB2 trigonal planar AB3 tetrahedral AB4 trigonal bipyramidal AB5 octahedral AB6 lone pairs occupy more space than bonding pairs electrons on the … PCl3 lewis structure, molecular geometry, bond angle, … What is the Lewis Structure for CO? - Chemistry Stack Exchange With three bonding pairs and one lone pair, the structure is … meeting across time zones Repulsions are minimized by directing each hydrogen atom and the lone pair to the corners of a tetrahedron. How many lone pairs are in pcl3 mean WebThere are four electron groups around nitrogen, three bonding pairs and one lone pair. WebIn the Lewis structure of PCl3, how many bonds and how many lone pairs are on the central atom? (Note: In this question a double or a triple bond would count as one bond.) … photography 4 bulb light box lamps 5500k 10.2: VSEPR Theory - The Five Basic Shapes - Chemistry LibreTexts
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
